It is a diprotic acid that can release two protons, but is only weakly acidic due to the strong O-H bonds. The chemical structure of carbonic acid is shown below, and it consists of a carboxyl group, and two hydroxyl groups. Precipitation reactions in nature can account for mineral formation in many cases, such as with undersea vents called black smokers that form metal sulfides. Its molecular formula is CH 2 O 3, and its molar mass is 62.03 g/mol. So of course H2CO3 changes into carbon dioxide and water… but why? you can read a really interesting here to find out! It is pretty mind-boggling haha.Ĭredits to the following webpages:, chemistryland. Fe 2+ (aq) + 2 OH (aq) Fe (OH)2 (s) Al 3+ (aq) + PO 43 (aq) AlPO 4 (s) Minerals are water-insoluble compounds. 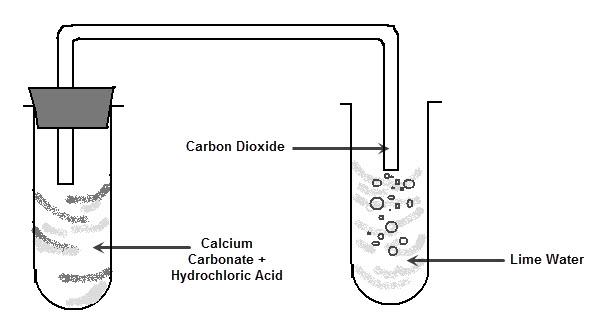
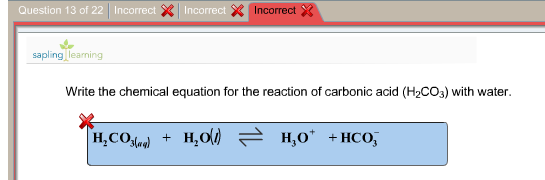
The picture above shows you that carbonic acid is H2CO3, it has 2H, 1C, and 3Os and how it is formed, umm the picture also shows a precipitation reaction btw, because calcium carbonate is insoluble and it will sort of precipitates out of solution by linking up with other calcium carbonate to form clumps.Īnyway, the key idea above for the baking soda reaction is that the real part of an acid that gives it acidic properties, which is the H+ ion is the one THAT REALLY REACTS at all, so the H+ ion reacts with the part of bicarbonate that is gives it basic properties which is actually the HCO3 to form voila! H2CO3 ABSTRACT: Wellbore integrity of abandoned wells is of high priority for ensuring the containment of sequestered CO2. The carbonic acid decomposes to water and carbon dioxide gas. When water is added, the acid and bicarbonate combine to make a salt and carbonic acid. Baking powder is a mixture of acetic acid and baking soda. If the 25.00 mL sample that was titrated had been produced by dilution of a 10.YEAH! acid and bases may sound like martians or other kind of aliens, but hey look at this, it is inolved in the baking of everyday’s good old bread!īaking soda is sodium bicarbonate.If 37.63 mL of thiosulfate solution were needed to titrate a 25.00 mL sample of an acid, what was the H + ion concentration of the acid? The thiosulfate solution was determined to be 1.023 M.The endpoint is reached when the solution becomes colorless. An example of a precipitation reaction is given below: Both reactants are aqueous and one product is solid. The amount of iodine produced from this reaction can be determined by subsequent titration with thiosulfate: 2S 2O 3 2+ + I 2 → S 4O 6 2− + 2I − A double replacement reaction is specifically classified as a precipitation reaction when the chemical equation in question occurs in aqueous solution and one of the of the products formed is insoluble. Reactions (a) Mixing an aqueous solution produces precipitate (b) mixing two colorless solutions produces a blue solution, 5. Reactions (a) heating copper in air produces a black solid (b) heating copper in vacuum produce - red-brown film, 3. Consequently, pH is increased around the cell and induces precipitation of calcium carbonate in presence of soluble Ca 2+ (Equations 57). Study with Quizlet and memorize flashcards containing terms like 1. Starch is used as the indicator in this titration because starch reacts with iodine in the presence of iodide to form an intense blue complex. Carbonic acid is converted to bicarbonate (Equation 3) by carbonic anhydrase (CA) and two moles of ammonium and hydroxide are formed due to ammonia hydrolysis (Equation 4). ♦ A method for determining the molarity of a strongly acidic solution has been developed based on the fact that a standard solution of potassium iodide and potassium iodate yields iodine when treated with acid: IO 3 − + 5I − + 6H + → 3I 2 + 3H 2O This equation does not result any net change in water hardness. If the overall reaction sequence was 75% efficient, how many grams of carbonic acid were initially dissolved in the 250 mL to produce the calcium bicarbonate? Eq.(1) is the neutralization reaction between CO 2 carbonic acid and lime.If 8.5 g of calcium carbonate were obtained by evaporating 250 mL of a solution of calcium bicarbonate followed by drying, what was the molarity of the initial calcium bicarbonate solution, assuming complete reaction?.The reprecipitated calcium carbonate forms a hard scale, or incrustation, on the surface of the object. This weakly acidic carbonic acid solution dissolves marble, converting it to soluble calcium bicarbonate: CaCO 3(s) + H 2CO 3(aq) → Ca(HCO 3) 2(aq)Įvaporation of water causes carbon dioxide to be driven off, resulting in the precipitation of calcium carbonate: Ca(HCO 3) 2(aq) → CaCO 3(s) + H 2O(l) + CO 2(g) CaAl 2 Si 2 O 8 + H 2 CO 3 + O 2 -> Al 2 Si 2 O 5 (OH) 4 + Ca 2+ + CO 32- plagioclase + carbonic acid -> kaolinite + dissolved calcium + carbonate ions This reaction shows calcium plagioclase feldspar, but similar reactions could also be written for sodium or potassium feldspars. Groundwater dissolves atmospheric carbon dioxide to produce an aqueous solution of carbonic acid: CO 2(g) + H 2O(l) → H 2CO 3(aq) 
♦ Reactions that affect buried marble artifacts present a problem for archaeological chemists. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
